-
-
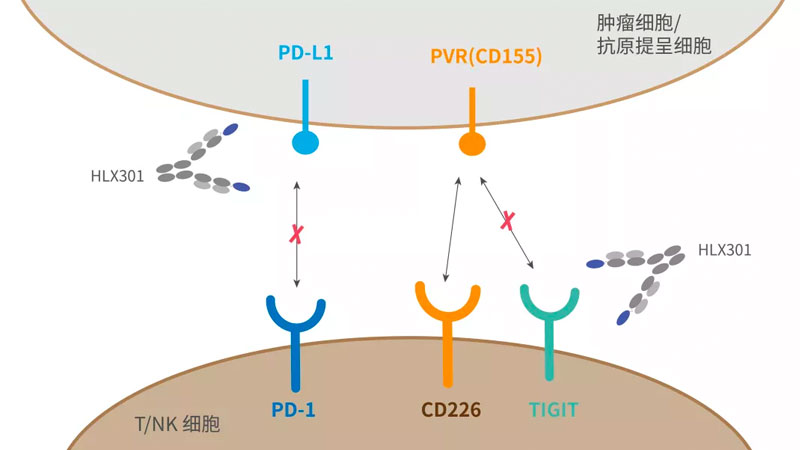
- 2021-11-25
Henlius Novel Anti-PD-L1/TIGIT Bispecific Antibody Receives Clinical Trial Approval in Australia
Shanghai, China, Nov, 24th, 2021 - Shanghai Henlius Biotech, Inc. (2696.HK) announced that the filing of a clinical trial for HLX301, a Recombinant Humanized Anti-PDL1 and Anti-TIGIT Bispecific Antibody
-
-
-
- 2021-11-12
Raw Materials and Preparations of Moxifloxacin Hydrochloride of Guilin Pharma Approved for Marketing and Passes Consistency Evaluation
On November 3, 2021, the raw materials and preparations (strength: 0.4 g) of moxifloxacin hydrochloride produced by Guilin Pharma were approved for marketing, which was deemed to have passed the consistency evaluation.
-
-
-

- 2021-11-09
Successful Completion of Phase I Clinical Trial of Pertuzumab Biosimilar HLX11 of Henlius
Shanghai, China, November 8th, 2021-Shanghai Henlius Biotech, Inc. (2696.HK) announced a phase 1 clinical trial of the company’s pertuzumab biosimilar HLX11, a recombinant anti-human epidermal growth factor receptor 2 (HER2) subdomain II humanized monoclonal antibody injection
-
-
-
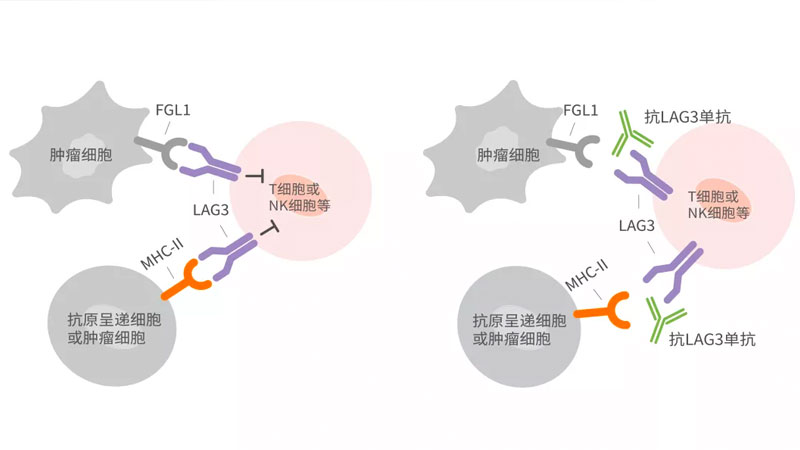
- 2021-10-13
First Subject Dosed in Phase I Clinical Trial of Henlius Novel Monoclonal Antibody HLX26, Inhibitor of New Generation of Immune Checkpoint LAG-3
Shanghai, China, October 12th, 2021 – Shanghai Henlius Biotech, Inc. (2696.HK) announced that the first subject has been dosed with HLX26 (recombinant anti-LAG-3 human monoclonal antibody injection) for the Phase 1 clinical trial in patients with advanced/metastatic solid tumours or lymphomas.
-
-
-

- 2021-09-14
Yi Bao (Human Erythropoietin for Injection) for Cancer-related Anemia Indication Officially Approved
On September 14, 2021, Wanbang Biopharma announced that the new indication of Yi Bao (Human Erythropoietin for Injection) was recently officially approved by the National Medical Products Administration (NMPA) for the treatment of anemia caused by chemotherapy in patients with non-myeloid malignancies.
-
-
-

- 2021-08-18
HAN QU YOU® (60 mg/bottle) Officially Approved for Marketing for More Flexible Medication
On August 17, 2021, Henlius (2696. HK) announced that the Company had recently received the Approval Letter of Supplementary Application issued by the National Medical Products Administration (NMPA): HAN QU YOU® (trastuzumab, Zercepac in the EU) was formally approved by the NMPA for the sNDA of new strength (60 mg/vial, free of preservatives). After adding a new strength of 60 mg/vial in China, patients can use different strengths of HAN QU YOU® for more flexible medication.
-
-
-

- 2021-08-17
Axicabtagene Ciloleucel of Fosun Kite Included in the Breakthrough Therapeutic Drug Program
Fosun Kite Biotechnology Co., Ltd. announced today that the National Medical Products Administration (NMPA) has officially included the new indication of Axicabtagene Ciloleucel, a CD19-targeted autologous CAR-T cell therapy product of Fosun Kite, into the Breakthrough Therapy Designation (BTD) for the treatment of relapsed or refractory indolent non-Hodgkin's lymphoma (r/r iNHL), including follicular lymphoma (FL) and marginal zone lymphoma (MZL), after two or more lines of systemic therapy.
-
-
-
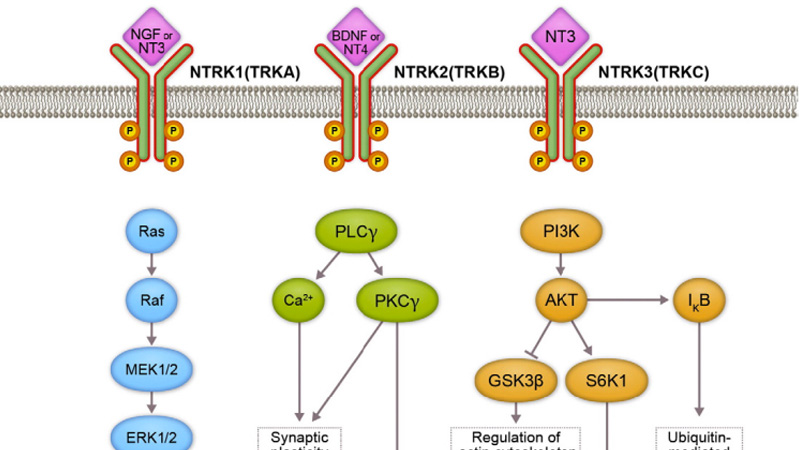
- 2021-06-28
Fosun Pharma: Second-generation TRK Inhibitor FCN-098 Clinically Approved
Recently, the Investigational New Drug Application (IND) was approved for FCN-098 capsules, a Class 1 new drug independently developed by Chongqing Fochon Pharmaceutical Research Co., Ltd. (hereinafter referred to as "Fochon Pharmaceuticals"). As a second-generation TRK inhibitor, FCN-098 is intended for the treatment of patients with advanced solid tumors with NTRK gene mutation.
-
-
-

- 2021-06-24
Fosun Pharma: Metronidazole Tablets of Guilin Pharma, Subsidiary of Fosun Pharma, Passes Generics Consistency Evaluation
On June 17, Guilin Pharmaceutical Co., Ltd. received the Approval Letter of Supplementary Application of metronidazole tablets issued by the National Medical Products Administration (NMPA): the drug passed generics consistency evaluation. Allegedly, metronidazole tablets manufactured by only 7 pharmaceutical companies in China, including Guilin Pharma, have passed the consistency evaluation.
-
-
Our Company
Shanghai Fosun Pharmaceutical (Group) Co., Ltd. is a global innovation-driven pharmaceutical and healthcare industry group deep-rooted in China

- Fosun Pharma is patient-centered and clinical needs-oriented. The company continuously enriches its innovative product pipeline through independent research and development, cooperative development, license-in, and in-depth incubation. Fosun Pharma improves the research and clinical development capabilities of FIC (First-in-class) and BIC (Best-in-class) new drugs as well as accelerates the R&D and launch of innovative technologies and products.
-
Our Business
Shanghai Fosun Pharmaceutical (Group) Co., Ltd. (Fosun Pharma) directly operates businesses including pharmaceutical manufacturing, medical devices, medical diagnosis, and healthcare services. As a shareholder of Sinopharm Group Co., Ltd., Fosun Pharma expands its areas in the pharmaceutical distribution and retail business.

- Fosun Pharma is patient-centered and clinical needs-oriented. The company enriches its innovative product pipeline through diversified and multi-level cooperation models such as independent research and development, cooperative development, license-in, and in-depth incubation. Fosun Pharma has formed technological platforms for innovative small molecule drugs, antibody drugs, and cell therapy with a focus on key disease areas including oncology and immunomodulation, metabolism and digestive system, and central nervous system. Fosun Pharma also vigorously explores cutting-edge technologies, such as RNA, oncolytic viruses, gene therapy and PROTAC, to enhance its innovation capabilities.
-
R&D
Shanghai Fosun Pharmaceutical (Group) Co., Ltd. is a global innovation-driven pharmaceutical and healthcare industry group deep-rooted in China.

- Guided by innovation and internationalization, the company continuously enriches its innovative product pipeline through diversified and multi-level cooperation models such as independent research and development, cooperative development, license-in, and in-depth incubation. Fosun Pharma improves the research and clinical development capabilities of FIC (First-in-class) and BIC (Best-in-class) new drugs as well as accelerates the R&D and launch of innovative technologies and products.
-
Sustainability
In pursuit of sustainable development of talents and products is the social responsibility concept of Fosun Pharma.

- Fosun Pharma has incorporated sustainable development into the Group's overall development strategy, emphasizing long-term ESG governance capabilities, and established a three-level ESG governance structure supervised by the board of directors (the “Board”), implemented by the ESG Committee, and executed by the ESG Working Group. The ESG Committee of the Board is responsible for formulating and promoting the Group's ESG vision, goals, and strategies, and provides advice to the Board; the ESG Working Group is responsible for sorting out key ESG issues, formulating sustainable development quantitative targets, and tracking progress. The ESG Committee and the ESG Working Group are committed to integrating ESG concepts into corporate operations and enhancing the company's sustainability.
-
Investor Relations
Shanghai Fosun Pharmaceutical (Group) Co., Ltd. is a global innovation-driven pharmaceutical and healthcare industry group deep-rooted in China.

- Founded in 1994, Shanghai Fosun Pharmaceutical (Group) Co., Ltd. ("Fosun Pharma"; stock code: 600196.SH, 02196.HK) is a global innovation-driven pharmaceutical and healthcare industry group deep-rooted in China. Fosun Pharma directly operates businesses including pharmaceutical manufacturing, medical devices, medical diagnosis, and healthcare services. As a shareholder of Sinopharm Group Co., Ltd., Fosun Pharma expands its areas in the pharmaceutical distribution and retail business.
-
News
Shanghai Fosun Pharmaceutical (Group) Co., Ltd. is a global innovation-driven pharmaceutical and healthcare industry group deep-rooted in China.

- Founded in 1994, Shanghai Fosun Pharmaceutical (Group) Co., Ltd. ("Fosun Pharma"; stock code: 600196. SH, 02196. HK) is a global innovation-driven pharmaceutical and healthcare industry group deep-rooted in China. Fosun Pharma directly operates businesses including pharmaceutical manufacturing, medical devices, medical diagnosis, and healthcare services. As a shareholder of Sinopharm Co., Ltd., Fosun Pharma expands its areas in the pharmaceutical distribution and retail business.
-
Careers
Some top talent with strong entrepreneurship gathers here with us.

- We are looking for people that recognize and practice our cultural values, and can learn fast to create continuous value; possess profound professional knowledge of the industry, a wide vision of different regions, and global-level expertise in a certain field. We will also be committed to cultivating international top talent with outstanding performance and great potential.


